Reacciones de transferencia de electrones. Autoevaluación
Tabla de potenciales normales
|
|
Semirreacciones |
Potencial estándar de reducción E°(V) |
|||
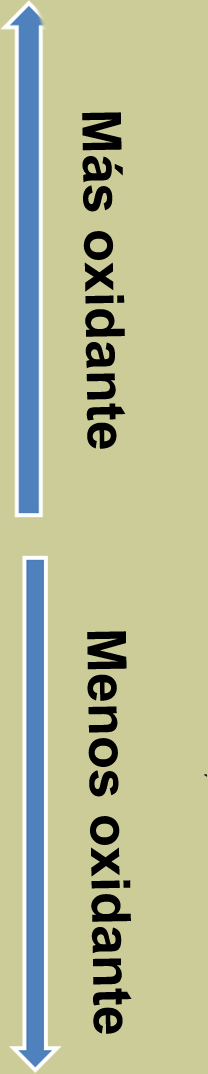 |
F2 + 2e- |
2 F- | 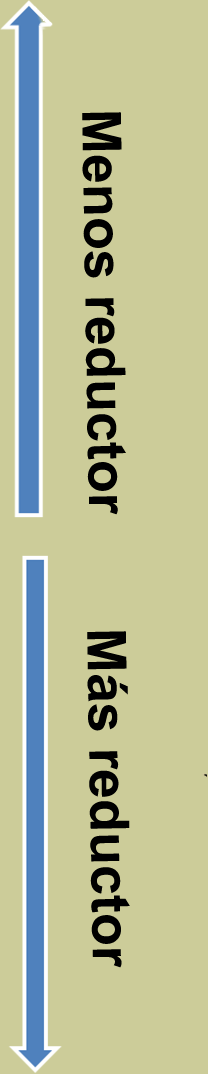 |
+2.87 | |
|
Co3+ + 1e- |
Co2+ | +1.82 | |||
|
H2O2 + 2 H+ + 2e- |
2 H2O |
+1.78 | |||
|
MnO4-+ 8H++ 5e- |
|
Mn2+ + 4 H2O |
+1.49 |
||
|
Cl2(g) + 2e- |
|
2Cl- |
+1.36 |
||
|
Cr2O72- + 14H+ + 6e- |
|
2Cr3+ + 7H2O |
+1.33 |
||
|
O2 + 4H+ + 4e- |
|
2H2O |
+1.23 |
||
|
IO3- + 6H+ + 5e- |
|
1/2 I2 + 3H2O |
+1.20 |
||
|
Br2 + 2e- |
2Br - | +1.06 | |||
|
NO3- + 4H+ +3e- |
|
NO+ 2H2O |
+0.96 | ||
|
NO3- + 3H+ +2e- |
|
HNO2 + H2O |
+0.94 |
||
|
Cu2+ + I- + 1e- |
|
CuI |
+0.86 |
||
Hg22+ + 2e- |
2Hg | +0.80 |
|||
|
Ag+ + 1e- |
|
Ag |
+0.80 |
||
|
Fe3+ + 1e- |
|
Fe2+ |
+0.77 |
||
|
2HgCl42- + 2e- |
|
Hg2Cl2(s) + 6Cl- |
+0.69 |
||
|
I2 (aq) + 2e- |
|
2I- |
+0.62 |
||
|
I3- + 2e- |
|
3I- |
+0.54 |
||
|
AsO43-+ 2H++ 2e- |
AsO33-+ H2O | +0.58 | |||
|
SO32- + 6H+ + 4e- |
S + 3H2O | +0.45 | |||
|
O2(g) + 2H2O + 4e- |
4OH- | +0.40 | |||
|
Cu2+ + 2e- |
|
Cu(s) |
+0.34 |
||
|
Hg2Cl2(s) + 2e- |
|
2Hg + 2Cl- |
+0.27 |
||
|
AgCl + 1e- |
|
Ag + Cl- |
+0.22 |
||
|
Sn4+ + 2e- |
|
Sn2+ |
+0.15 |
||
|
Cu2+ + 1e- |
|
Cu+ |
+0.15 |
||
|
S + 2 H+ + 2e- |
H2S |
+0.14 | |||
|
HCHO(aq) + 2H+ + 2e− |
CH3OH(aq) | +0.13 | |||
|
H2O2+ 2e- |
2OH- |
+0.12 | |||
|
S4O62- + 2e- |
|
2S2O32- |
+0.08 |
||
|
2H+ + 2e- |
|
H2 |
0.00 |
||
|
CrO42- +4H2O + 3e- |
|
Cr3+ + 8OH- |
-0.13 | ||
|
Sn2+ + 2e- |
|
Sn |
-0.14 |
||
|
2CO2 (g) + 2H+ + 2e- |
|
H2C2O4 |
-0.49 |
||
|
Zn2+ + 2e- |
|
Zn |
-0.76 |
||
|
2H2O + 2e- |
H2 + 2OH- | -0.88 | |||
|
Ca2+ + 2e− |
Ca(s) | −2.76 | |||
|
Li+ + 1 e- |
Li | -3.04 | |||
Este artículo está licenciado bajo Creative Commons Attribution-NonCommercial 2.5 License